“Annex 1 provides general guidance that should be used in the design and control of facilities, equipment, systems and procedures used for the manufacture of all sterile products applying the principles of Quality Risk Management (QRM), to ensure that microbial, particulate and endotoxin/pyrogen contamination is prevented in the final product”.
Continuous viable air monitoring in grade A (e.g. air sampling or settle plates) should be undertaken for the full duration of critical processing, including equipment (aseptic setup) assembly and critical processing. A similar approach should be considered for Grade B cleanrooms based on the risk of impact on the aseptic processing. The monitoring should be performed in such a way that all interventions, transient events and any system deterioration would be captured and any risk caused by interventions of the monitoring operations be avoided.






Aseptic Sterile Manufacturing .The art of performing a specific sterile manufacturing process within an ultra clean cleanroom environment to ensure that the product’s such as an injectable solution is transported from bulk sterile containers and filled into vials, or syringes or capsules. Ensuring complete sterility of the product.
EU GMP Annex 1 States
Continuous viable air monitoring in grade A (e.g. air sampling or settle plates) should be undertaken for the full duration of critical processing, including equipment (aseptic setup) assembly and critical processing. A similar approach should be considered for Grade B cleanrooms based on the risk of impact on the aseptic processing. The monitoring should be performed in such a way that all interventions, transient events and any system deterioration would be captured and any risk caused by interventions of the monitoring operations be avoided.
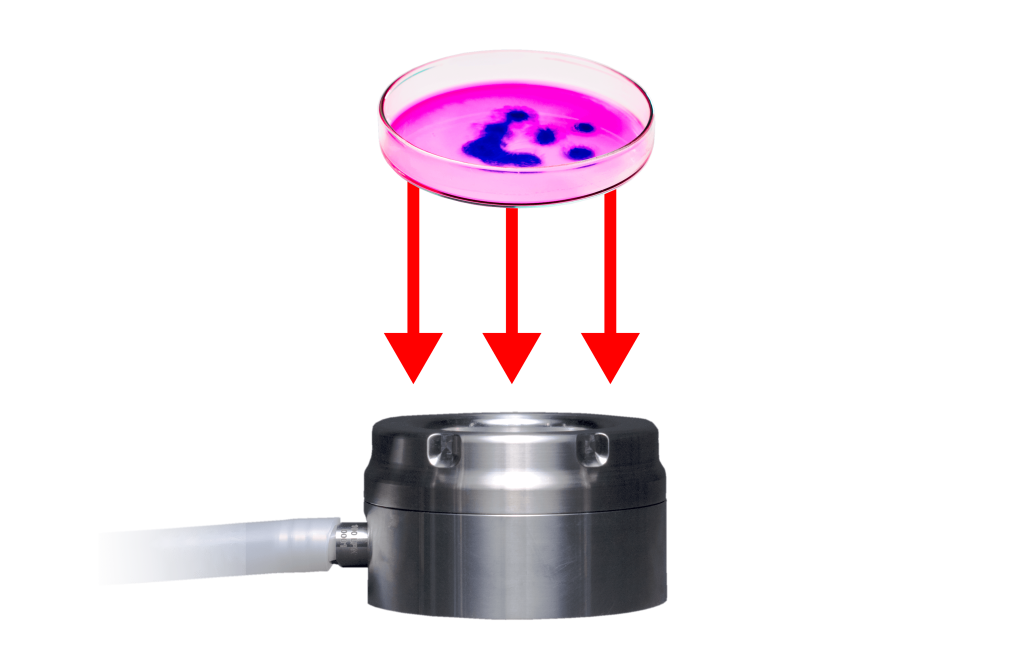
How do you meet continuous viable air monitoring? The best way is to understand the technology we use and to select the right viable monitoring solutions. Check out this quick video to get an idea of how to monitor a BSC using particle counters, active air samplers and settle plates.
“Annex 1 provides general guidance that should be used in the design and control of facilities, equipment, systems and procedures used for the manufacture of all sterile products applying the principles of Quality Risk Management (QRM), to ensure that microbial, particulate and endotoxin/pyrogen contamination is prevented in the final product”.
Lets help you understand how you can meet Annex1 continuous viable monitoring requirements using existing technology that you may have without breaking the bank or having to revalidate your system.[link to webinar]
Active air sampling during aseptic processing is a critical requirement in Grade A cleanrooms. Key considerations include the physical and biological efficiencies of air samplers, appropriate flowrates, and HEPA-filtered exhausts. The goal is to maintain an environment with zero CFUs
Surface swabbing is an essential method for collecting samples from various surfaces, aiding in trend analysis and verifying control procedures. Regular testing helps identify the presence of bacteria, yeast, and mold, ensuring the effectiveness of sterility measures.